|
The atomic number (expressed by the letter Z) of an element is the number of protons in the nucleus of each atom of that element. Objects, like asteriods or meteors, might have very different isotope quantities. The relative atomic masses specified in periodic table entries-for example, hydrogen-are calculated for all the naturally occurring isotopes of every element, weighted by the quantity of those isotopes on earth. The relative atomic mass is an average of all the atomic masses of different isotopes in a given sample, with each isotope's contribution to the average. Since an element’s isotope differs in its atomic masses, scientists may also establish the relative atomic mass at times called the atomic weight for an element. All isotopes of a particular element have exactly the same number of protons but again only differ in the number of neutrons in each atom. In general, an atom's atomic mass will be very close to its mass number but will have some difference in the decimal places.Īn isotope of an element is a variant of that particular element that consists of a difference in the number of neutrons and further in the nucleon number. By definition, an atom of carbon with 6 neutrons, carbon-12, has an atomic mass of 12 amu. The atomic mass of a single atom is basically its total mass and is usually expressed in atomic mass units or amu. 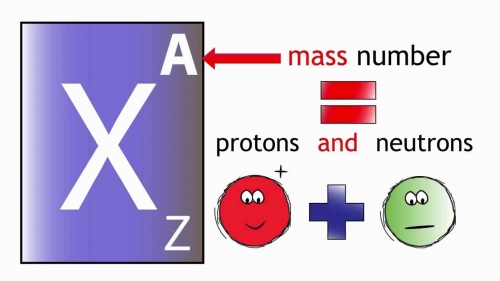
The notation of an atom is given below.Ī property strongly related to an atom’s mass number is its atomic mass. If you want to calculate the number of neutrons in an atom, you can simply deduct the number of protons, or atomic numbers, from the mass number. Mass Number = Number of protons + Number of neutrons Together, the number of protons and the number of neutrons dictates an element’s mass number. On the other hand, the number of neutrons for a given element can differ. The atomic number is the number of protons there in an atom. In fact, the number of protons dictates what atom we are looking at (e.g., all atoms with 6 protons are carbon atoms). Here, we will discuss important concepts of atomic number and atomic mass.Ītomic Number, Atomic Mass, and Relative Atomic MassĪtoms of each element consist of a particular number of protons. The atomic number and mass number are the basic concepts of atomic structure.  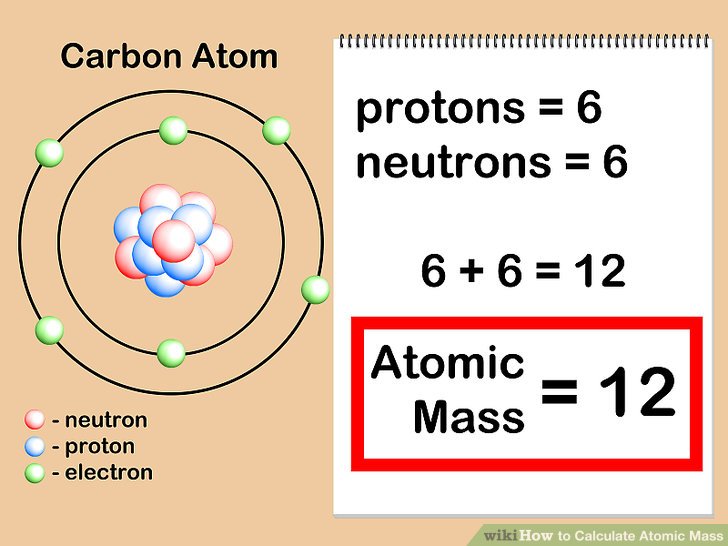
The mass number is defined as the total number of protons and neutrons of an element. Hence atomic number is useful in differentiating one element from another. No element has an atomic weight less than one.The atomic number is defined as the total number of protons or electrons in an element. One atomic mass unit isĭefined as 1/12 the mass of an atom of carbon-12. 
The number in parentheses is the mass number of the most stable or best-known isotope of that element.Ītomic weights are measured in atomic mass units. These elements are unstable that is, their nuclei decompose radioactively. The atomic weight of some elements is given as a whole number enclosed in parentheses. That 75.53% of naturally occurring chlorine is chlorine-35 and 24.47% is chlorine-37. The atomic weight of chlorine (35.45) reflects the fact The atomic weight of carbon (12.01) reflects the relative abundance of The atomic weight of an element is based on the variety of naturally occurring isotopes of that element and the relative abundance of each.Ī collection of naturally occurring carbon atoms contains 98.89% carbon-12Ītoms and 1.11% carbon-13 atoms, along with a trace percentage of carbon-14Ītoms. Both the periodic table and the alphabetical list of the elements show the atomic weights of the elements. The atomic weight (or atomic mass) of an element is the average relative mass of the naturally occurring atoms of that element.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
